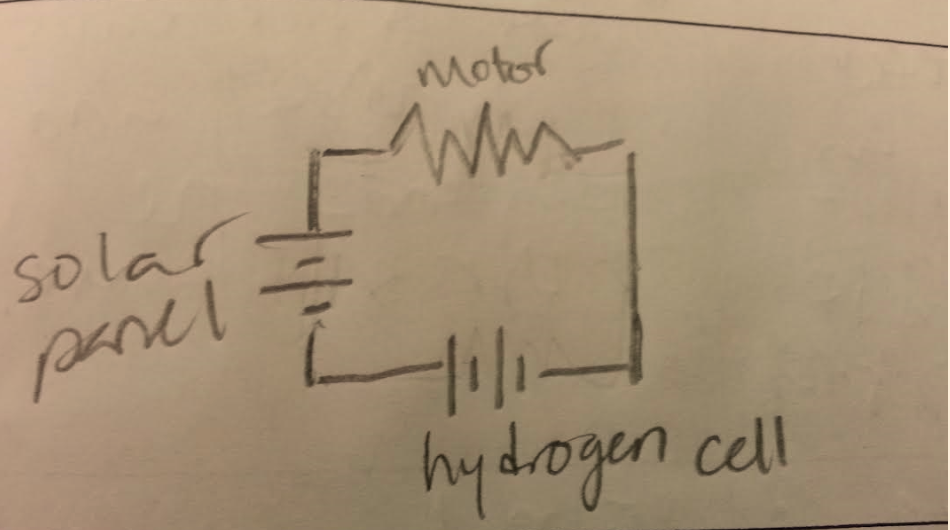
Solar Hydrogen System
Hydrogen Cells
Hydrogen cells may be the fuel of the future, but they were discovered over 175 years ago. They were first discovered by accident during an electrolysis experiment in 1839 by Sir William Grove. However this model was impracticable. It wasn't until the 1930s that fuel cells were researched again. In 1950, Francis Bacon created the first Alkaline fuel cell, which was much more efficient than Grove's cell. In the 1960s, NASA modified Bacon's fuel cell for the Apollo missions. Nowadays there are 6 main types of fuel cells- Alkaline, Polymer Electrolyte Membrane, Phosphoric Acid, Molten Carbonate, Solid Oxide, and Direct Methanol. The cells combine hydrogen and oxygen to create electricity, water, and heat.
Solar Panels
Solar panels utilize rays of light from the sun or other powerful light sources that can be converted into energy which then can create electricity. Solar cells use materials that give off electrons when hit by photons of light which is called the photoelectric effect. The solar panel is able to capture the free electrons and convert them to an electric current, or electricity, through these materials and electrical conductors. This electricity is then used to power houses and buildings. This type of power is very environmentally friendly and because it is powered by the sun, it is an inexhaustible resource.
Series and parallel circuits
There are two main types of circuits- series and parallel. A series circuit is a circuit that only has one path for the current to flow. This means that if the path is broken anywhere in the circuit, none of the components will work. There are also parallel circuits, which have more than one path for the current to flow through. Because of this, if the circuit is broken, the system can still function because there is another path for the current to take. Parallel and series circuits can also be put together to create combination circuits, which have components from both types of circuits.
Circuit
Reflection
I enjoyed this project and I thought that we did pretty well, however I think we could have had better time management. Our main problem was that in order to do calculations we had to charge our hydrogen and solar cells, which in some circumstances, didn't even work in the first place. This took at least 15 minutes, which in an hour long class period, is a pretty big chunk of time. So I just wish that we could have had our cells started for us earlier. Another one of our minor problems was getting the wheel and axle to work. Also, several of our work days some of our group members were missing, which made the progression of our project slow down. Our motor would run, but because of the gears we used in our original prototype, there was too much torque for the motor to overcome, so the axle wouldn't spin. It was only once we got rid of the gear system completely that the axle was able to move.
Activity 1.3.1
1. Read the Fuel Cell User Guide.
2. Follow the directions in the Fuel Cell User Guide under the section Preparing the Fuel Cell for Use.
3. Shine a bright light source on the solar panel, always keeping at least 8 inches of separation between the two to avoid melting the solar module plastic.
Set your multimeter to measure voltage and connect the multimeter test leads to the solar panel terminals. Move the solar panel or light source to determine the location that produces the highest voltage value. You may want to mark the positions with some tape. Record the open-circuit voltage. Note the current is zero, since a voltmeter has nearly infinite resistance.
VOC = Open-Circuit Voltage 1.5V Power = VOC x 0 A = 0 W
4. With the test leads disconnected, set your multimeter to measure current. Return the solar module to the same exact position that produced the highest voltage value and measure the current. Record this short-circuit current. Note that the voltage is zero, since an ammeter has nearly zero resistance.
ISC = Short-Circuit Current 114mA Power = 0 V x ISC = 0 W
5. Calculate the amount of power that would be produced by the solar module if it could simultaneously produce the voltage and current you measured in the previous two steps.
For this illumination level, the solar module will deliver, at most, about 70% of this theoretical maximum, and will do so at a resistance between zero and infinite resistance.
Maximum Theoretical Power = VOC x ISC = 0.171 W
6. Attach the solar panel to the solar hydrogen automobile. Using a standoff or another suitable method, prop up one end of the chassis so that the motor-driven wheel is not in contact with the ground. Connect the motor leads to the solar module using the breadboard to make the connections. Position the light source to produce maximum voltage leaving a minimum distance of 8 inches between solar module and the lamp. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
7. Set your multimeter to measure voltage. Connect the multimeter test leads to the solar module terminals. Record the load voltage value.(Drive gear should be engaged)
V = Load Voltage 2V
8. Disconnect the test leads and set your multimeter to measure current. Connect the multimeter in series with the solar module. Record the load current.
I = Load Current = 200mA
9. Calculate the power delivered by the solar module when it is loaded by the motor with the wheels off the ground.
P = Load Power = I V = 400mW for solar module.
10. Energize the fuel cell by using one of the power sources according to the directions in the Fuel Cell User Guide under the section Powering the Fuel Cell (Electrolysis).
Fuel cells can be damaged by high current. If using a DC power supply with the Heliocentris fuel cell, do not use more than 500 mA. Do not use a battery to energize the fuel cell.
11. After the fuel cell is energized, attach the fuel cell to the motor using the breadboard to make the connections. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
12. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the fuel cell terminals. Record the voltage value.
V = Load Voltage 0.2 V
13. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the fuel cell.
Caution! Never measure current from the fuel cell without a resistor, motor, or other load in series with the ammeter. Doing so can permanently damage the fuel cell.
Record the current value. Load Current = 30mA
14. Calculate the power delivered by the fuel cell. P = Load Power = I V = 6mW for fuel cell.
15. Remove the fuel cell and solar module and attach the two AAA battery holders to your vehicle using zip ties. Using the breadboard, connect the batteries in series with each other and with the motor. (See next step for wiring hints.) Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
16. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage 3V
17. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = __________
18. Calculate the power delivered by the batteries in series. P = Load Power = I V = __________ for batteries in series
19. Using the breadboard, connect the batteries in parallel with each other and with the motor. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? ______
20. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage __________
21. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = __________
22. Calculate the power delivered by the batteries in parallel.
P = Load Power = I V = __________ for batteries in parallel
1. Read the Fuel Cell User Guide.
2. Follow the directions in the Fuel Cell User Guide under the section Preparing the Fuel Cell for Use.
3. Shine a bright light source on the solar panel, always keeping at least 8 inches of separation between the two to avoid melting the solar module plastic.
Set your multimeter to measure voltage and connect the multimeter test leads to the solar panel terminals. Move the solar panel or light source to determine the location that produces the highest voltage value. You may want to mark the positions with some tape. Record the open-circuit voltage. Note the current is zero, since a voltmeter has nearly infinite resistance.
VOC = Open-Circuit Voltage 1.5V Power = VOC x 0 A = 0 W
4. With the test leads disconnected, set your multimeter to measure current. Return the solar module to the same exact position that produced the highest voltage value and measure the current. Record this short-circuit current. Note that the voltage is zero, since an ammeter has nearly zero resistance.
ISC = Short-Circuit Current 114mA Power = 0 V x ISC = 0 W
5. Calculate the amount of power that would be produced by the solar module if it could simultaneously produce the voltage and current you measured in the previous two steps.
For this illumination level, the solar module will deliver, at most, about 70% of this theoretical maximum, and will do so at a resistance between zero and infinite resistance.
Maximum Theoretical Power = VOC x ISC = 0.171 W
6. Attach the solar panel to the solar hydrogen automobile. Using a standoff or another suitable method, prop up one end of the chassis so that the motor-driven wheel is not in contact with the ground. Connect the motor leads to the solar module using the breadboard to make the connections. Position the light source to produce maximum voltage leaving a minimum distance of 8 inches between solar module and the lamp. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
7. Set your multimeter to measure voltage. Connect the multimeter test leads to the solar module terminals. Record the load voltage value.(Drive gear should be engaged)
V = Load Voltage 2V
8. Disconnect the test leads and set your multimeter to measure current. Connect the multimeter in series with the solar module. Record the load current.
I = Load Current = 200mA
9. Calculate the power delivered by the solar module when it is loaded by the motor with the wheels off the ground.
P = Load Power = I V = 400mW for solar module.
10. Energize the fuel cell by using one of the power sources according to the directions in the Fuel Cell User Guide under the section Powering the Fuel Cell (Electrolysis).
Fuel cells can be damaged by high current. If using a DC power supply with the Heliocentris fuel cell, do not use more than 500 mA. Do not use a battery to energize the fuel cell.
11. After the fuel cell is energized, attach the fuel cell to the motor using the breadboard to make the connections. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
12. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the fuel cell terminals. Record the voltage value.
V = Load Voltage 0.2 V
13. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the fuel cell.
Caution! Never measure current from the fuel cell without a resistor, motor, or other load in series with the ammeter. Doing so can permanently damage the fuel cell.
Record the current value. Load Current = 30mA
14. Calculate the power delivered by the fuel cell. P = Load Power = I V = 6mW for fuel cell.
15. Remove the fuel cell and solar module and attach the two AAA battery holders to your vehicle using zip ties. Using the breadboard, connect the batteries in series with each other and with the motor. (See next step for wiring hints.) Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? Yes
16. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage 3V
17. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = __________
18. Calculate the power delivered by the batteries in series. P = Load Power = I V = __________ for batteries in series
19. Using the breadboard, connect the batteries in parallel with each other and with the motor. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? ______
20. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage __________
21. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = __________
22. Calculate the power delivered by the batteries in parallel.
P = Load Power = I V = __________ for batteries in parallel
Conclusion
1. Using the measurements you made, compare and relate the four options you explored. Was the car best powered by a single fuel cell, a single solar module, two AAA batteries in series, or two AAA batteries in parallel?
Because of time constraints my group was not able to complete the calculations to answer this question, but based on the calculations I was able to complete, I would say the two AAA batteries in a series.
2. Did voltage, current, or power best describe the suitability of a power source?
I would say that power best describes the suitablility of a power source because power is the rate at which work is preformed in time. Because our goal was to create the fastest car, I think that power best describes the suitability of a power source.
3. If you had many solar modules, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the solar modules in terms of parallel and series circuits.
Refer to question 1.
4. If you had many fuel cells, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the fuel cells in terms of parallel and series circuits.
Refer to question 1.
5. Describe and defend a system that you believe would best utilize a solar hydrogen system to meet the needs for an average driver.
I think that the best way to use a solar hydrogen system to meet the needs of an average driver would be to have solar panels installed on the roof and a hydrogen cell stored somewhere where it can remain upright and safe, like in a special compartment in the trunk of the car. The car would also need to be able to store solar power for during drives at night or in overcast weather. This system would allow for maximum exposure to sunlight and would keep the hydrogen cell safe so it can work.
6. How does a photovoltaic cell work? Record the source of your information.
Photovoltaic cells take advantage of the photoelectric effect, which is a property that causes some materials to release electrons when they absorb photons of light. When these electrons are captured they create an electric current that can be used as electricity. Photovoltaic cells are made of thin semiconductor cells that are specially treated create an electric field, positive on one side and negative on the other. When photons of light strike the semiconductor wafer, the photoelectric effect occurs, and if electrical conductors are attached to the positive and negative sides of the wafer, the electrons can be captured and can create an electric current.
http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7. Detail how electrolysis separates hydrogen and oxygen. How is electricity produced as the fuel cell allows the hydrogen to reunite in a bond with oxygen? Record the source of your information.
Water is made of two positive hydrogen ions and one negative oxygen ion. When an electric current is run through the water via electrodes, the positive hydrogen ions are attracted to and collected by the negative electrode, which also neutralizes
the ion . The same happens with the negative oxygen ion and the positive electrode.
http://www.instructables.com/id/Separate-Hydrogen-and-Oxygen-from-Water-Through-El/
When the hydrogen and oxygen are reunited, the reaction results in about 0.7 V of electricity.
http://auto.howstuffworks.com/fuel-efficiency/alternative-fuels/fuel-cell2.htm
Because of time constraints my group was not able to complete the calculations to answer this question, but based on the calculations I was able to complete, I would say the two AAA batteries in a series.
2. Did voltage, current, or power best describe the suitability of a power source?
I would say that power best describes the suitablility of a power source because power is the rate at which work is preformed in time. Because our goal was to create the fastest car, I think that power best describes the suitability of a power source.
3. If you had many solar modules, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the solar modules in terms of parallel and series circuits.
Refer to question 1.
4. If you had many fuel cells, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the fuel cells in terms of parallel and series circuits.
Refer to question 1.
5. Describe and defend a system that you believe would best utilize a solar hydrogen system to meet the needs for an average driver.
I think that the best way to use a solar hydrogen system to meet the needs of an average driver would be to have solar panels installed on the roof and a hydrogen cell stored somewhere where it can remain upright and safe, like in a special compartment in the trunk of the car. The car would also need to be able to store solar power for during drives at night or in overcast weather. This system would allow for maximum exposure to sunlight and would keep the hydrogen cell safe so it can work.
6. How does a photovoltaic cell work? Record the source of your information.
Photovoltaic cells take advantage of the photoelectric effect, which is a property that causes some materials to release electrons when they absorb photons of light. When these electrons are captured they create an electric current that can be used as electricity. Photovoltaic cells are made of thin semiconductor cells that are specially treated create an electric field, positive on one side and negative on the other. When photons of light strike the semiconductor wafer, the photoelectric effect occurs, and if electrical conductors are attached to the positive and negative sides of the wafer, the electrons can be captured and can create an electric current.
http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7. Detail how electrolysis separates hydrogen and oxygen. How is electricity produced as the fuel cell allows the hydrogen to reunite in a bond with oxygen? Record the source of your information.
Water is made of two positive hydrogen ions and one negative oxygen ion. When an electric current is run through the water via electrodes, the positive hydrogen ions are attracted to and collected by the negative electrode, which also neutralizes
the ion . The same happens with the negative oxygen ion and the positive electrode.
http://www.instructables.com/id/Separate-Hydrogen-and-Oxygen-from-Water-Through-El/
When the hydrogen and oxygen are reunited, the reaction results in about 0.7 V of electricity.
http://auto.howstuffworks.com/fuel-efficiency/alternative-fuels/fuel-cell2.htm